 
Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. The chemistry of iron makes it a key component in the proper functioning of red blood cells. Transition metals have interesting chemical properties, partially because some of their electrons are in d subshells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The first element appearing on the list that is not a main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. Most periodic tables provide additional data (such as atomic mass) in a box that contains each element’s symbol. A modern version is shown in Figure 1.12.1 1.12. (Make sure you visit this, because I have mentioned the valence electrons along with the images for each element). The periodic table is one of the cornerstones of chemistry because it organizes all the known elements on the basis of their chemical properties. Elements are either metals, nonmetals, or semimetals. But if you want to see the valence electrons of all the 118 elements, then visit: Valence electrons of all the elements. Valence Electron – Wikipedia.\): Types of Elements. It is important to note that for both transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. The electrons of incomplete d-orbitals may also be considered as valence electrons in some inner transition metals. Similarly, for inner transition elements (or f-block elements), the valence electrons can range from 3 to 16 due to the contribution of both the f-subshell and s-subshell electrons. 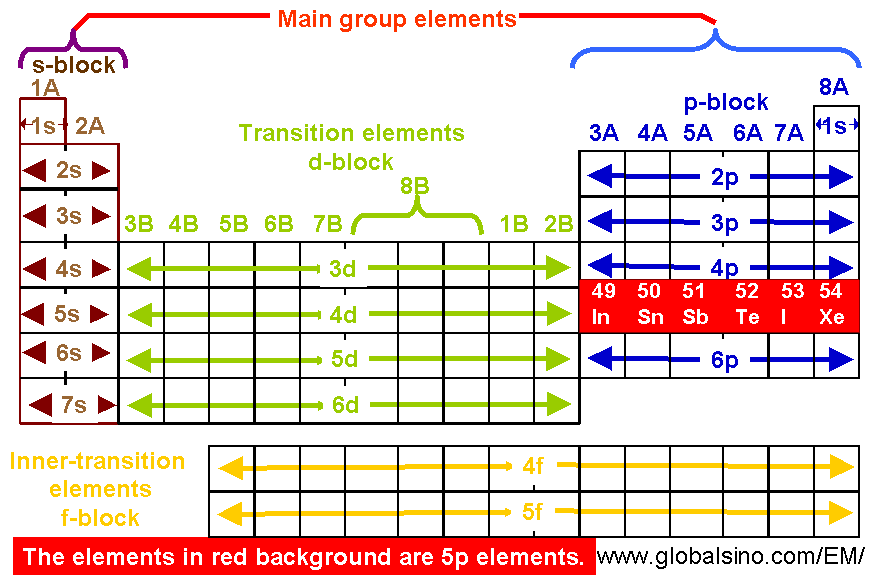
In some cases, the electrons of incomplete d-orbitals are also considered as valence electrons. This is because the d-subshell of transition elements and f-subshell of inner transition elements are incompletely filled and are very close to the outer s-subshell.Īs a result, the electrons of both the d-subshell and s-subshell in transition elements and f-subshell and s-subshell in inner transition elements can behave like valence electrons.įor transition elements, the valence electrons can range from 3 to 12 due to the contribution of both the d-subshell and s-subshell electrons. Valence electrons of transition elements and inner transition elements are more complicated to determine than those of main group elements. Valence electrons of transition and inner-transition elements These valence electrons are responsible for the elements’ unique chemical properties and reactivity. However, one might think that since the number. Electron affinity decreases down the groups and from right to left across the periods on the periodic table because the electrons are placed in a higher energy level far from the nucleus, thus a decrease from its pull. The number of valence electrons in these elements is equal to their group number.įor example, elements in group 1, such as lithium and sodium, have one valence electron, while elements in group 2, such as magnesium and calcium, have two valence electrons.Įlements in group 13, such as boron and aluminum, have three valence electrons, and so on. The less valence electrons an atom has, the least likely it will gain electrons. The main group elements, also known as the representative elements, are located in groups 1, 2, and 13-18 of the periodic table. The number of valence electrons an atom has determines its position in the periodic table and its ability to form chemical bonds. These electrons are responsible for the chemical properties of an element, such as its reactivity and the types of chemical bonds it can form with other atoms. Valence electrons are the electrons located in the outermost shell or energy level of an atom that are involved in chemical bonding.įor example, magnesium has 2 electrons in its outermost orbit, hence it has 2 valence electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
